|
Craft beers have become wildly successful over the last few years and one reason consumers are so crazy about them is that key ingredient – hops. Hops are a plant added during the beer making process to impart a bitter flavour to balance out sweetness, and add unique tropical or citrus aromas or flavours. But all those darn hipsters and their hoppy beer means the global supply of hops can’t meet the demand, making them very expensive. On top of that, they require a lot of water and natural resources to grow. Furthermore, the actual content of the essential oils within the hops can vary wildly, so it’s hard to achieve a consistent hoppy taste in beer. Researchers at the University of California – Berkeley have genetically engineered a strain of brewer’s yeast that means you can make hoppy beer without the hops.
First they identified two particular chemical compounds that give hoppy beer its special taste and smell, and then they found enzymes (biological catalysts) that can make these compounds from precursors already produced naturally by the yeast. They used CRISPR/Cas9 gene editing technology (you know, those molecular scissors I talked about in a previous post?) to genetically engineer yeast to produce high levels of these flavour molecules. This is a pretty impressive achievement because firstly, brewer’s yeast, unlike domesticated wine yeast, is pretty wild and has lots of crazy things going on with its chromosomes, making it notoriously difficult to genetically manipulate. Secondly, making sure those genetic manipulations stick so that the population is stable is pretty challenging. The researchers then used these new strains of yeast to ferment beers and checked that the levels of the flavour molecules were consistent. They showed much less variation than traditional dry-hopped beers, which will help brewers ensure a consistent taste. But, as they always say, the proof of the beer is in the drinking. A panel of taste testers consistently rated the beers as having a hoppy flavour and aroma. Of course, the complex flavour of hops cannot be completely encapsulated by just two molecules, but these methodologies provide a foundation to build more complex yeast strains that produce a wider variety of flavours and aromas. Ultimately, this technology means not only can you brew hoppy beer without the hops, but it paves the way for further improvements in brewer’s yeast and advancements in environmental sustainability.
0 Comments
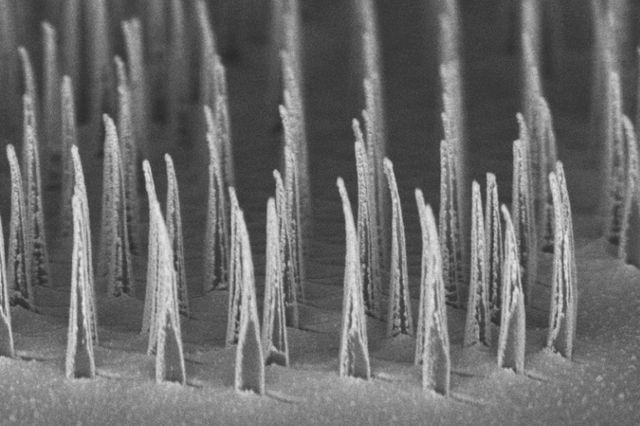
Many human diseases such as cystic fibrosis, muscular dystrophy, AIDS, and cancer are caused by disruptions to genes. Gene therapy is an emerging technology that aims to replace missing or defective genes within human cells to treat these conditions. However, it is currently still very risky and not widely tested on humans. Existing approaches to gene therapy use viruses, electric fields or harsh chemicals. These may pose a risk to the cell in terms of toxicity or undesirable off-target effects, as well as being expensive and potentially inefficient. Scientists are currently working on ways to make gene therapy safer, faster, cheaper and more efficient. One of the major challenges in delivering gene therapy is making sure the therapeutic agents can target the desired areas and enter the cells. The trick is to get the therapeutics to enter the cell without killing it or affecting its growth. A group of researchers at the University of California – Los Angeles have developed tiny ‘nanospears’ made of silicon, nickel and gold. Because the tips of these spears are so infinitesimally small they are able to penetrate the membrane of the cell without harming it. Nanospears aren’t new – other groups have worked on similar structures powered by nanoscale motors in order to propel the spears towards their target. However, these newly-developed spears are even better as they are magnetic, so scientists can use a magnetic field to guide the nanospears without having to use toxic chemicals. They are also bio-degradable and can be mass-produced, making them cheaply available on a large-scale.
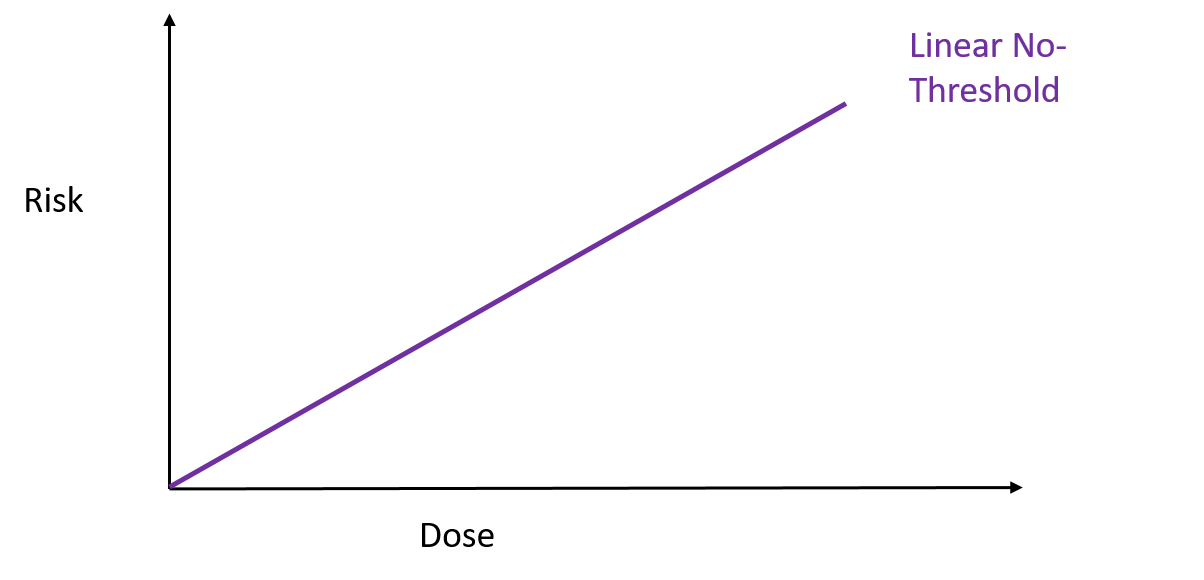
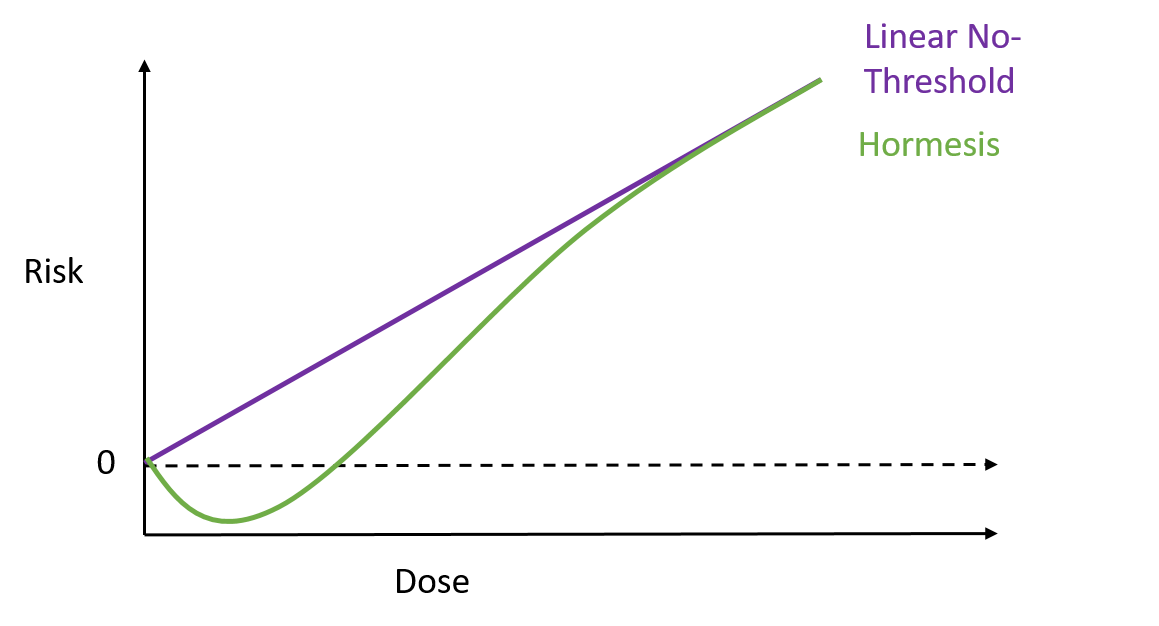
To test how well these nanospears worked, the researchers used them to deliver a gene for green fluorescent protein – that’s right, it does exactly what it says on the label – into a culture of human cells. Approximately 80% of the cells produced a green glow, indicating the gene reached its target, and of those cells 90% survived. That’s a marked improvement over current forms of gene therapy. So far this technique has been used to deliver DNA to cell culture systems. Not only is this an extremely a useful tool for researchers, but it may also help speed up the delivery of gene therapy and immunotherapy. But that’s not all. These nanospears could be used to deliver more than just DNA, paving the way for new nanomedicine technologies that could be used for applications ranging from biological research to human medicine. The researchers are currently working on optimising the nanospears technology and hope that in the future it could be used to guide the delivery of gene therapies within a human patient. Hey everyone, this week's blog is a bit different to normal because it's based on a body of research rather than a single recent paper. I'll admit, I got a bit behind this week so I've recycled a post from my Sci Comm class last year. It's a little longer than normal but well worth the read in my opinion - let me know what you think! Radiation can be a pretty scary thing. After all, the effects of an overdose of radiation can be devastating. Radiation overexposure can cause radiation sickness, burns, cataracts, infertility and cancer. Some of these effects can take years to manifest. The reason radiation is so dangerous is because it is able to damage cells by disrupting the chemical bonds between molecules inside the body. If a cell is greatly damaged it will die, and the death of many cells can lead to radiation sickness and even death. Alternatively, if radiation damages a cell’s DNA it can cause it to mutate and become cancerous. But radiation is an everyday part of life. On a daily basis we are exposed to radiation from space, air, rocks, buildings, food, and even from our own body. Radioactive elements are everywhere, and there’s no way of avoiding them completely. What matters is the dose Generally we think of radiation exposure as directly dose dependent -- that is, any radiation is harmful, with no safe level of exposure. This is called the linear no-threshold model and this is the model used by regulators and policy-makers to determine radiation guidelines. However, there’s evidence that this might be wrong. While there’s no question that high levels of radiation leads to a greater cancer risk, there’s less data about how low levels of exposure might affect this risk. This is partly because our natural incidence of cancer is fairly high and can vary greatly depending on lifestyle factors, making it hard to untangle the exact effects of chronic low-dose radiation. Despite this, there are some studies that show exactly the opposite of what you would expect: that low doses of radiation might be good for you. In Taiwan, during the 1980s, a batch of recycled steel was accidentally contaminated with radioactive cobalt. This steel was used to make over 180 buildings containing roughly 1700 apartments. Approximately 10,000 residents of the apartments were exposed to higher than normal levels of radiation over 9-20 years. Within this group of residents, the cancer mortality rate was much lower than in the general population – in fact, it was only 3% of the expected rate. The rate of congenital birth defects was also significantly lower – only 6.5% of the expected rate for the general population. This is possibly the best example of a case study on low-dose radiation because the sample size is large, and other factors such as lifestyle and environment are likely to be consistent between the sample population (the residents) and the general population of Taiwan. But it is not the only example. A number of other studies have shown that people living in areas with naturally higher background radiation have a lower cancer mortality rate than the general population. So, it appears chronic low doses of radiation may actually lower the risk of cancer. This phenomenon is termed ‘radiation hormesis’. It’s not certain why radiation hormesis may exist, but several explanations have been proposed.
It’s possible that radiation hormesis is an adaptive response to damage. Cell exposed to a very low dose of radiation, given a few hours to recover, and then exposed to a higher dose of radiation are less likely to sustain DNA damage than unexposed cells. This means cells that are pre-exposed are more able to protect themselves against higher levels of radiation. Other effects of low dose radiation include:
Animal studies have shown support for radiation hormesis. One experiment using mice predisposed to cancer found low doses of radiation helped slow down the rate of cancer growth. However, analysis of data in humans suggests a mixture of effects. Phillipe Duport, the founder of the Centre for Low Dose Research at the University of Ottowa says “A little radiation may be good for some people but bad for others.” Some scientists believe there is already enough evidence to accept the radiation hormesis hypothesis, and go so far as to say the linear no-threshold model is ‘scientifically unfounded’. Others believe proponents of the hormesis theory have chosen to ignore works that support the linear no-threshold model. However, most of the data supporting the linear no-threshold model is from survivors of the Japanese atomic bomb. These results may be complicated by other factors, such as exposure to cancer-causing chemicals released during the bombing. The conflicting evidence demonstrates a need for further research on humans, but this poses an obvious ethical dilemma. A strong experimental design would require a large number of subjects willing to be exposed to radiation over a long period of time. Unfortunately such a study is unlikely to be approved by an ethics committee, so we may have to rely on data from ‘happy accidents’ like the Taiwan apartments. Currently, advisory bodies like the National Council on Radiation Protection and Measurements are sticking to the linear no-threshold model. On one hand, acceptance of the hormesis model would redefine ‘safe’ levels of radiation exposure, and potentially save millions of dollars in the clean-up of radiation accidents. On the other, regulators tend to err on the side of caution, and mistakes in this area could be devastating to the lives of the people affected. Nevertheless, there’s stacks of evidence to support radiation hormesis, and pressure is growing on policy makers. So perhaps we will see a change sometime soon. And maybe you won’t run away screaming next time you see a radiation sticker. Genome editing is a vital tool for scientists to be able to understand diseases, improve crops, and unlock the secrets of how our cells work, as well as many other applications. Up until about 5 years ago, genetic engineering usually meant weeks of time and effort, was expensive and had limited versatility. Now, similar experiments can be conducted in less than a week very, very cheaply. So, what changed? The introduction of CRISPR/Cas9 technology revolutionised genome editing. CRISPR (clustered regularly interspaced short palindromic repeats) meant that genes could be knocked out (deleted), inserted, or mutated easily, with high accuracy, at almost any position in the genome. Based off of a bacterial immune defence system, the CRISPR/Cas9 complex consists of several components. The Cas9 protein forms a complex with guide RNA that matches the sequence of the DNA to be targeted. The complex scans the genome for the protospacer adjacent motif (PAM) sequence, which is a short series of DNA bases immediately after the targeted DNA sequence. When it finds a match, the guide RNA pairs with the target DNA sequence and the Cas9 protein cuts the DNA in half, forming a double stranded break. Double stranded breaks in DNA are bad news for the cell, so the cell attempts to repair the cut. However, this process is prone to errors, so often mutations are introduced which means the gene no longer functions. Alternatively, scientists can introduce new genes by adding DNA which is integrated at the site of the cut. In this way, scientists can add or delete genes to study how they work. CRISPR/Cas9 is very versatile and efficient, but it’s not perfect. While it can target almost any region of the genome, it still needs that PAM sequence. The most commonly used protein recognises NGG (N being any DNA base and G being guanine), which is relatively short but still has to be present in the target sequence. CRISPR/Cas9 is also limited by off-target effects. It may accidentally cut other regions of the genome that closely match the guide RNA sequence and introduce unwanted mutations.
Recently, a group of researchers at Harvard set out to improve the Cas9 protein. They used a technique that forced the protein to rapidly evolve and accept a wider range of PAM sequences. They tested this new xCas9 variant with mammalian cells for compatibility and found that it will be suitable for a broad range of applications, especially in human cells. Surprisingly, xCas9 has greater specificity for the target DNA sequence, with fewer off-target effects observed than with the original Cas9. This illustrates that there is not necessarily a trade-off between specificity and versatility, and may help to improve future generations of Cas9 proteins. This improvement means CRISPR/Cas9 genome editing is even more versatile and will be able to target regions of the genome more specifically and more precisely, expanding the scope of genome editing and enhancing the potential of using CRISPR technology to understand and treat human disease. |
Author
Emi Schutz Archives
March 2018
Categories |




 RSS Feed
RSS Feed
